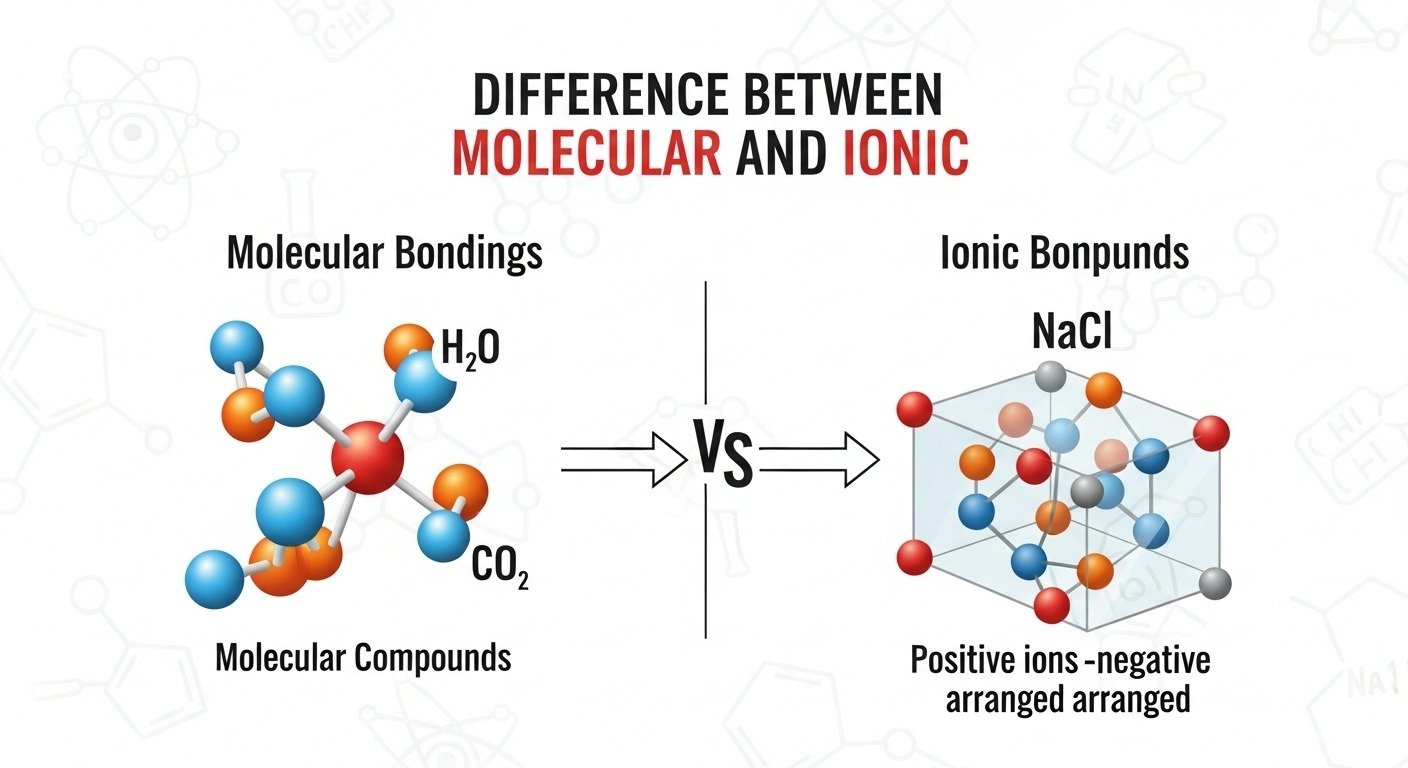
A student once tried dissolving table salt and sugar in water during a chemistry experiment. Both disappeared, but their behavior was completely different. This simple moment reveals the difference between molecular and ionic compounds. Chemistry hides fascinating contrasts in everyday materials.
Molecular compounds are substances where atoms share electrons to form covalent bonds. On the other hand, ionic compounds are created when atoms transfer electrons, forming charged particles called ions. This basic concept explains the difference between molecular and ionic substances found in food, medicine, and technology.
Understanding the difference between molecular and ionic helps learners see how materials behave in water, electricity, and chemical reactions. Small atomic changes create big differences in properties.
Scientists and students study the difference between molecular and ionic compounds to predict melting points, conductivity, and reactivity. This knowledge powers chemistry education and real-world innovation.
In short, the difference between molecular and ionic explains why sugar tastes sweet but salt conducts electricity in solution. Chemistry becomes clearer when we see these distinctions.
Key Difference Between the Both
The main difference between molecular and ionic compounds lies in how atoms bond together.
- Molecular compounds form through sharing electrons between non-metal atoms, creating covalent bonds.
- Ionic compounds form when electrons transfer from a metal to a non-metal, producing positively and negatively charged ions.
This bonding difference changes their structure, conductivity, melting point, and solubility.
Why Their Difference Is Necessary to Know for Learners and Experts
Understanding the difference between molecular and ionic compounds is crucial in chemistry, medicine, engineering, and environmental science.
Students learn why some substances dissolve easily while others conduct electricity. Chemists use this knowledge to design medicines, batteries, and industrial chemicals.
In society, ionic compounds help produce electrolytes, salts, and fertilizers, while molecular compounds are essential for life molecules like water, oxygen, and carbon dioxide.
Knowing this difference allows experts to predict chemical reactions and create safer materials.
Pronunciation
Molecular
- US: /məˈlɛk.jə.lɚ/
- UK: /məˈlek.jʊ.lə/
Ionic
- US: /aɪˈɑː.nɪk/
- UK: /aɪˈɒn.ɪk/
Linking Hook
Now that we understand the basic concept and importance, let’s explore the detailed difference between molecular and ionic compounds through clear comparisons and practical examples.
Difference Between the Keywords
1. Type of Bond
Molecular compounds form through covalent bonds, where atoms share electrons.
Examples: Water (H₂O), Carbon dioxide (CO₂)
Ionic compounds form through ionic bonds, where electrons transfer between atoms.
Examples: Sodium chloride (NaCl), Calcium chloride (CaCl₂)
2. Elements Involved
Molecular compounds usually involve non-metals bonding with non-metals.
Examples: Methane (CH₄), Oxygen (O₂)
Ionic compounds involve metals and non-metals.
Examples: Sodium fluoride (NaF), Potassium bromide (KBr)
3. Structure
Molecular compounds form discrete molecules.
Examples: Ammonia (NH₃), Hydrogen peroxide (H₂O₂)
Ionic compounds form crystal lattice structures.
Examples: Sodium chloride crystal, Magnesium oxide (MgO)
4. Melting and Boiling Points
Molecular compounds usually have low melting and boiling points.
Examples: Carbon dioxide, Methane
Ionic compounds have high melting and boiling points.
Examples: Sodium chloride, Calcium oxide
5. Electrical Conductivity
Molecular compounds do not conduct electricity.
Examples: Sugar solution, Ethanol
Ionic compounds conduct electricity when dissolved or molten.
Examples: Saltwater solution, Molten sodium chloride
6. Solubility
Molecular compounds may dissolve depending on polarity.
Examples: Sugar dissolving in water, Oxygen dissolving in water
Ionic compounds often dissolve easily in water.
Examples: Sodium chloride, Potassium nitrate
7. Physical State
Molecular compounds are often gases or liquids at room temperature.
Examples: Oxygen gas, Water
Ionic compounds are usually solid crystals.
Examples: Table salt, Lithium fluoride
8. Bond Strength
Molecular bonds are generally weaker intermolecular forces.
Examples: Hydrogen bonding in water, van der Waals forces in methane
Ionic bonds are strong electrostatic attractions.
Examples: NaCl lattice, MgO crystal
9. Formation Process
Molecular compounds form when atoms share electrons for stability.
Examples: Hydrogen molecule, Nitrogen molecule
Ionic compounds form when atoms gain or lose electrons.
Examples: Sodium chloride, Aluminum oxide
10. Chemical Behavior
Molecular compounds often participate in biological and organic reactions.
Examples: Glucose reactions, DNA molecules
Ionic compounds often appear in electrolytes and salts.
Examples: Sodium chloride in food, Potassium ions in cells
Nature and Behaviour
Molecular Compounds
They behave as individual molecules with weaker intermolecular forces. They often form gases, liquids, or soft solids and participate in biological processes.
Ionic Compounds
They behave as crystalline solids with strong electrostatic forces. They are rigid, stable, and conduct electricity when dissolved.
Why People Are Confused About Their Use
People often confuse them because both look similar as solids or solutions in daily life. Sugar and salt both dissolve in water, but their internal bonding is completely different. Without chemical knowledge, the difference is not visible.
Table Showing Difference and Similarity
| Feature | Molecular Compounds | Ionic Compounds |
| Bond Type | Covalent | Ionic |
| Elements | Non-metal + Non-metal | Metal + Non-metal |
| Structure | Molecules | Crystal lattice |
| Melting Point | Low | High |
| Conductivity | Poor | Conducts in solution |
| Physical State | Gas/liquid/soft solid | Hard solid |
| Solubility | Variable | Usually soluble in water |
Similarity: Both form stable chemical substances and participate in chemical reactions.
Which Is Better in What Situation?
Molecular compounds are better for biological and organic systems. They form the basis of life molecules like proteins, water, and oxygen, making them essential for living organisms and biochemical reactions.
Ionic compounds are better for electrical conduction and industrial uses. They are widely used in batteries, salts, electrolytes, and minerals that support agriculture and human health.
Use in Metaphors and Similes
Though mostly scientific terms, they can appear metaphorically.
- “Their friendship was molecular, tightly sharing everything.”
- “The group formed an ionic bond, strongly attracted but with opposite personalities.”
Connotative Meaning
Molecular
- Connotation: Neutral/Positive
- Suggests complexity and scientific precision.
Example: “The research focused on molecular structures.”
Ionic
- Connotation: Neutral
- Suggests strong attraction or charged interaction.
Example: “Their debate had an ionic intensity.”
Idioms or Proverbs
These words rarely appear in traditional idioms, but science-inspired expressions exist:
- “Like an ionic bond” – describing strong attraction
Example: “Their teamwork was like an ionic bond—powerful and stable.” - “At a molecular level” – meaning very detailed
Example: “The scientist explained the problem at a molecular level.”
Works in Literature
Molecular
- The Molecular Biology of the Gene — Science, James Watson, 1965
- Molecular Biology of the Cell — Science textbook, Bruce Alberts, 1983
Ionic
- Ionic Processes in Chemistry — Chemistry reference, various authors, 1970s
Movies Related to the Keywords
Molecular / Molecular Science Themes
- The Nutty Professor (1996, USA) – chemistry transformation story
- Spider-Man (2002, USA) – molecular mutation science themes
Ionic / Chemical Themes
- Radioactive (2019, UK/France) – chemistry discoveries
- The Current War (2017, USA) – electricity and charged particles
Frequently Asked Questions
1. What is the main difference between molecular and ionic compounds?
Molecular compounds share electrons, while ionic compounds transfer electrons.
2. Do molecular compounds conduct electricity?
No, they usually do not conduct electricity.
3. Why do ionic compounds dissolve in water?
Water stabilizes ions, allowing them to separate and dissolve.
4. Are molecular compounds always gases?
No, they can be gases, liquids, or soft solids.
5. Which compound type has higher melting points?
Ionic compounds generally have higher melting points.
How Both Are Useful for Surroundings
Molecular compounds support life processes, air composition, and biological chemistry. Ionic compounds provide minerals, electrolytes, and industrial materials essential for agriculture, medicine, and energy systems.
Final Words for Both
Both molecular and ionic compounds form the foundation of chemical science. One represents shared electrons, while the other represents electron transfer and charged attraction.
Conclusion:
Chemistry becomes easier when we understand the difference between molecular and ionic compounds. These two types of substances differ mainly in how atoms bond sharing electrons or transferring them. This simple concept explains many everyday observations, from why salt conducts electricity to why sugar does not.
Molecular compounds consist of atoms sharing electrons to form covalent bonds. They often have lower melting points and play a huge role in biological systems. Life itself depends on molecular chemistry.
In contrast, ionic compounds form through electron transfer between metals and non-metals. The resulting charged ions create strong electrostatic forces that build solid crystal structures. These substances usually have high melting points and electrical conductivity in solution.
Understanding the difference between molecular and ionic helps students, scientists, and engineers predict chemical behavior and design useful materials. From medicines to batteries, this knowledge shapes modern science.
In the end, both types are equally important. Together they explain the structure, behavior, and diversity of the chemical world around us.

I am William Strunk Jr an American professor of English and the author of the classic style guide the Elements of Style. He emphasized clarity, brevity, and proper grammar in writing, influencing generations of writers. Learn more about his work and writing tips at wordrar.com.